Adipose Niches
Context of Use or Disease: Adipose niches in ovarian cancer cell migration to the omentum, adipose bone marrow niche
DOI: Nature Communications 2025, Biomaterials 2025
Platform: Compatible with multiple formats, including for microfluidic chip embedding and seeding of single cell suspensions or integration of spheroids and organoids
Description: These models use a microdroplet technology developed at QM allowing the control of adipocyte-mimicking droplet size, volume fraction/density and lipid composition, and integration within engineered matrices. This allows mimicking of important biochemical (matrix, growth factors, defined lipid compositions) and biophysical (mechanics and microstructure) features of adipose tissues, to model tumour development and resistance to therapies, or to recreate unique adipose niches regulating stem cell maintenance.
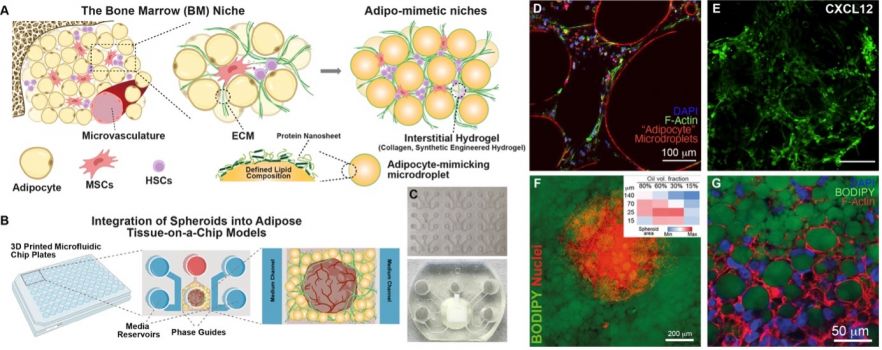
Fig. 1. A-B. Schematic representation of adipose tissue and bone marrow models and their embedding in chips. C. Images from microfluidic chip plates and single chip with embedded adipose tissue. D-E. Examples of confocal microscopy images of the bone marrow model. F-G. Examples of confocal microscopy images of the ovarian cancer spheroids integrated in adipose tissue models. Inset is a score of cancer cell migration and spheroid size as a function of “adipocyte” microdroplet size and volume fraction.
Characterisation & Validation: The control of adipo-mimetic tissue formation and control of multiscale mechanics, degradability, microstructure, lipid formulation and matrix biochemistry was demonstrated. The impact of adipose tissue engineering on ovarian cancer cell migration was characterised and validated against ex vivo samples (multiple cell lines) and patient progression. The maintenance of stem cells including mesenchymal stromal/stem cells (MSCs) and hematopoietic stem cells (HSCs) in adipose tissues was validated via qPCR, flow cytometry and multi-omics.
Ongoing Research: Deep modelling of the bone marrow HSC niche for fundamental studies and testing of safety/efficacy of therapeutics, investigation of complex therapeutics efficacy for ovarian cancer
Research Team: Julien Gautrot, Frances Balkwill, Paolo Gallipoli
Lead Contact: Julien Gautrot
Last updated 15/05/2026


