Glioma Outgrowth
Context of Use or Disease: Interactions between healthy and cancerous (glioma) brain tissue
DOI: In vitro models 2025
Platform: Photolithography moulded microfluidic chips on microscope slides
Description: A single channel, 3-well PDMS microfluidic chip is fabricated and mounted on glass coverslip. Healthy neural cells (CTX-TNA2 astrocyte cell line) are cultured to confluency, and a glioblastoma spheroid (9L/3cmv-GFP glioma cell line) is added into the centre well. Time course imaging can be used to measure cellular changes at the boundary of the cancerous growth into healthy neural tissue.
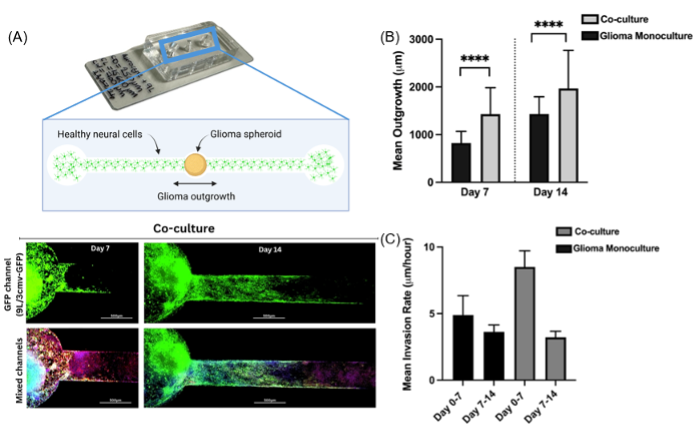
Fig. 1. A. Photo and fluorescence images of the glioma outgrowth model. B, C. Co-culturing of glioma with astrocytes resulted in significantly increased glioma outgrowth and higher initial invasion rate into the healthy tissue.
Characterisation & Validation: Linear channel enables quantification of outgrowth speed and distance. Outgrowth speed and distance were significantly increased when brain cancer cells were grown in co-culture with healthy neural cells, in line with recent advances in cancer neuroscience. Hallmark characteristics of invasive glioblastoma were seen, such as increases in astrocytic reactivity (GFAP) and glioma cell CD44 expression.
Ongoing Research: Transition to human induced pluripotent stem cell derived neural cultures and patient derived glioma organoids, fabrication of chips with 32 addressable electrodes for real-time electrophysiological recording along the entire microfluidic channel
Research Team: Christopher Chapman, Joshua Daoud
Lead Contact: Christopher Chapman
Last updated 16/04/2026


